
It's not just Maiden Pharmaceuticals. India's health care authorities also need to take the blame for the Gambian fiasco
Experts say lax inspection and certification processes mean substandard products by repeat offenders are allowed to be sold in domestic and export markets
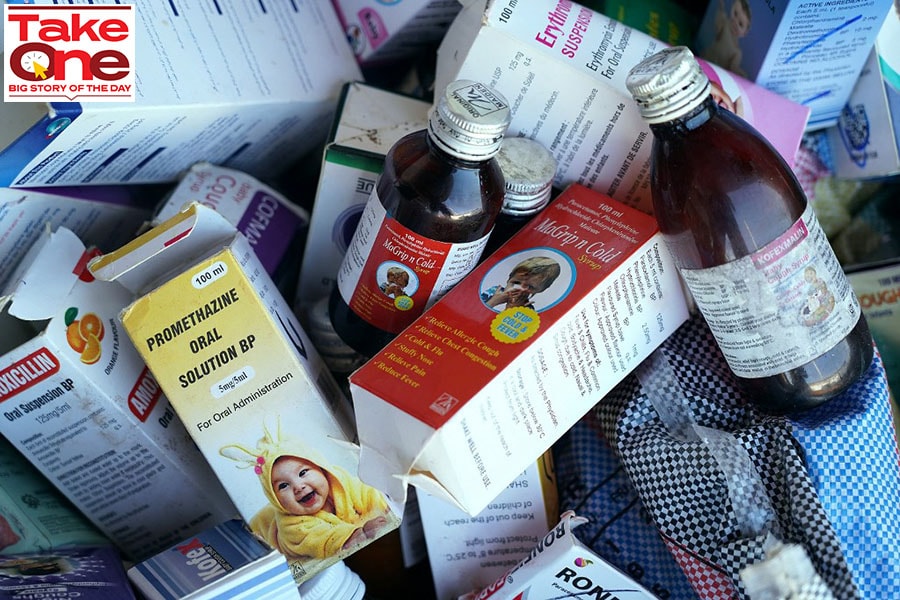 A photograph shows collected cough syrups in Banjul on October 06, 2022. - Indian authorities are investigating cough syrups made by a local pharmaceutical company after the World Health Organisation said they could be responsible for the deaths of 66 children in The Gambia.
Image: Milan Berckmans / AFP
A photograph shows collected cough syrups in Banjul on October 06, 2022. - Indian authorities are investigating cough syrups made by a local pharmaceutical company after the World Health Organisation said they could be responsible for the deaths of 66 children in The Gambia.
Image: Milan Berckmans / AFP
Up until October 5, New Delhi-headquartered Maiden Pharmaceuticals was having a relatively good run in the $25 billion pharmaceutical export market.
Maiden’s products were sold across the world, from Chile and Venezuela in the Latin American markets, to Senegal and Burkina Faso in Africa, and Vietnam and Cambodia in Southeast Asia. In all, according to the company’s website, its products are sold in over 41 countries.
On October 5, however, that dream run of 32 years came to a halt. The company is being accused of selling contaminated products that have been potentially linked to acute kidney injuries and 66 deaths among children in the Gambia, according to World Health Organization (WHO).
The four medicines are cough and cold syrups produced by Maiden: Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup, and Magrip N Cold Syrup. According to WHO, the company hasn’t to date provided guarantees to WHO on the safety and quality of the products.
Laboratory analysis of samples of each of the four Maiden products has confirmed, according to WHO, that they contain unacceptable amounts of diethylene glycol (DEG) and ethylene glycol as contaminants; WHO also reckons the products may have been sold outside of the Gambia.




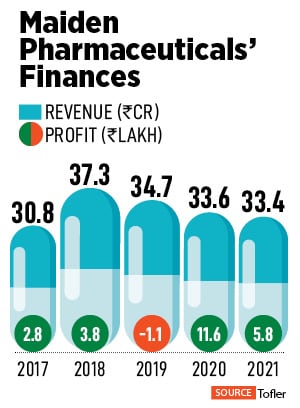 India has had a tumultuous history with DEG. Apart from the 12 children who died due to DEG poisoning in 2020, some 36 deaths were reported in Gurugram in 1998. Back then, too, children aged between 2 and 6 had died due to renal failure after consuming cough syrups made by a local manufacturer, according to news reports. Other instances include the death of 14 people in Mumbai in 1986 from DEG poisoning. In the US, the Federal Food, Drug, and Cosmetic Act was passed as a reaction to the death of 105 people who died from diethylene glycol poisoning in 1938.
India has had a tumultuous history with DEG. Apart from the 12 children who died due to DEG poisoning in 2020, some 36 deaths were reported in Gurugram in 1998. Back then, too, children aged between 2 and 6 had died due to renal failure after consuming cough syrups made by a local manufacturer, according to news reports. Other instances include the death of 14 people in Mumbai in 1986 from DEG poisoning. In the US, the Federal Food, Drug, and Cosmetic Act was passed as a reaction to the death of 105 people who died from diethylene glycol poisoning in 1938.



